The lab’s research projects are focused around the innate immune responses to virus infection in three main areas:
1) The innate immune response to viral nucleic acids.
Nucleic acids are powerful immunostimulatory agents. Sensing of damaged or mis-localised nucleic acids by pattern recognition receptors underlies multiple pathologies and is especially important in the context of anti-viral immunity.
Sensing of viral DNA in mammalian cells:
During DNA virus infection, the ability of our cells to sense viral genomic DNA it critical for mounting type one and type three interferon responses. Leveraging our expertise in poxvirology, a family of large DNA viruses which replicate their double stranded DNA genome in the cytoplasm, we discovered that mouse and human cells require DNA-dependent protein kinase (DNA-PK) for this process. DNA-PK binds to cytoplasmic DNA and activates the cGAS/STING pathway that drives the IRF-3 dependent production of interferons and cytokines in response to viral DNA. This pathway is required for mammalian cells to sense and respond to DNA viruses like herpesviruses and poxviruses. We also discovered that patients who have specific mutations in DNA-PKcs (the catalytic subunit of DNA-PK), can have enhanced STING signalling and IFN-I production, showing that this pathway functions in people and can lead to autoinflammatory pathology.
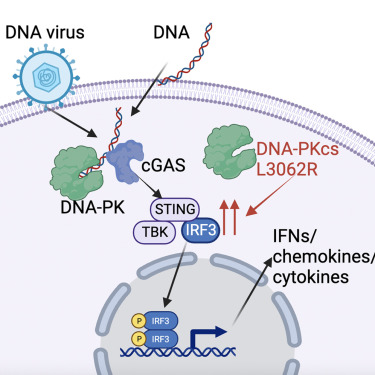
Sensing of viral RNA in mammalian cells:
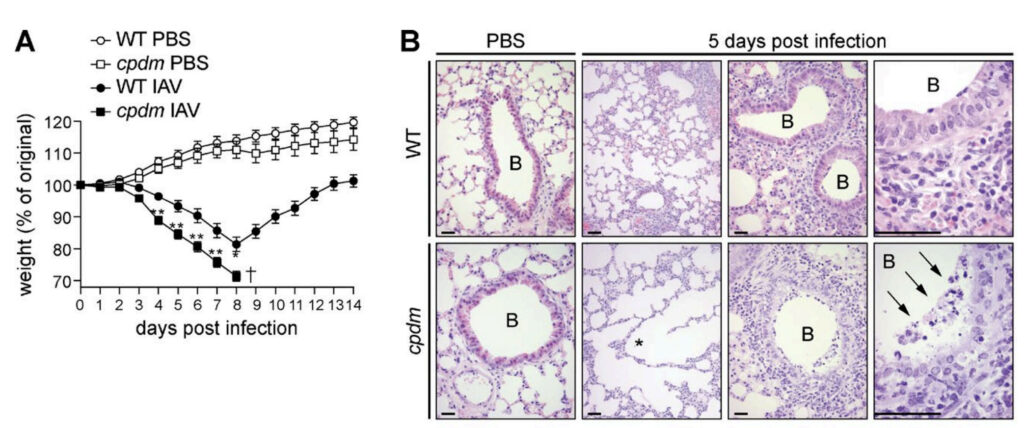
The receptors that respond to viral RNA, such as RIG-I, MDA5 and TLR3, are equally important for triggering mammalian interferon responses to RNA virus infections. We are using our understanding of cell signalling at the molecular, biochemical and cell biological level to determine the mechanisms by with RNA sensing receptors work. For example we have defined the contribution of the linear ubiquitin chain assembly complex (LUBAC) and M1-linked ubiquitin chains to anti-viral immunity via the regulation of RNA sensing pattern recognition receptors, TLR3 and RIG-I in the context of influenza, Zika and Sendai virus infection. For example, cpdm mice, that lack SHARPIN are more susceptible to influenza infection:
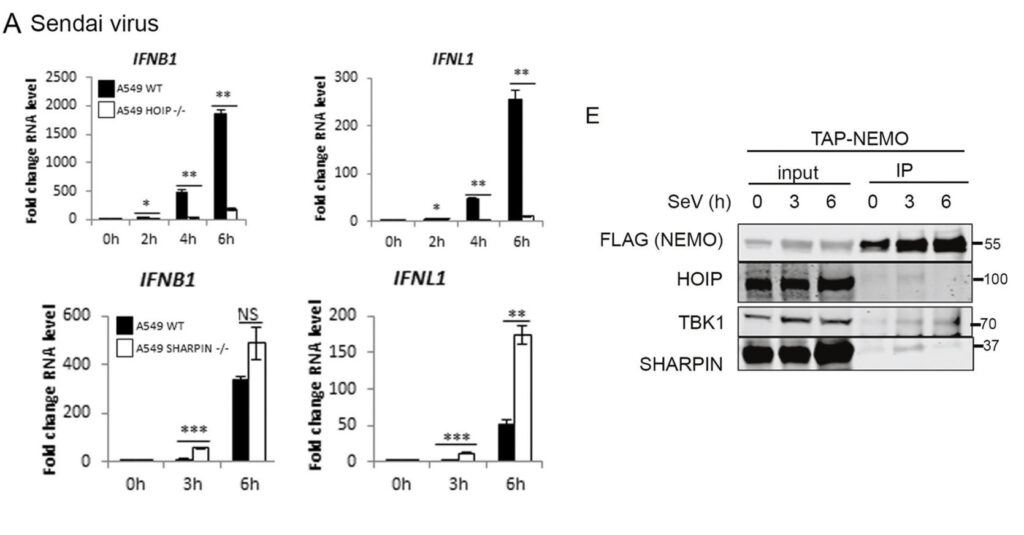
We also found differing roles for SHARPIN and HOIP (the catalytic component of LUBAC) in regulation of RIG-I signalling, with HOIP and HOIL-1 being required for antiviral RIG-I signalling but SHARPIN not, despite it being recruited to the RIG-I signalling complex via NEMO:
2) Evolution of poxvirus host/pathogen interactions
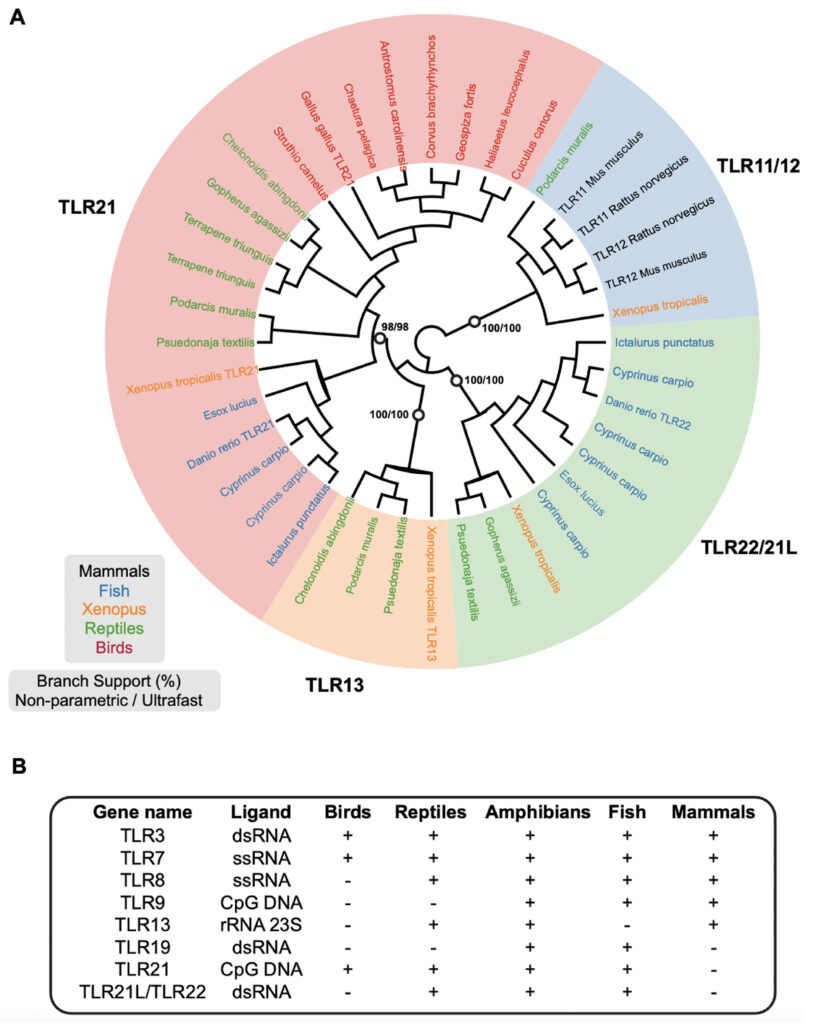
In to understand how nucleic acid sensing mechanisms evolved to drive anti-viral immunity we are taking a comparative immunology approach. We are defining nucleic acid sensing mechanisms in multiple species including humans, mice and birds and, in parallel, defining how poxviruses subvert these pathways to optimise their replicative niches and driving evolution of host defence mechanisms. We have show, for example, that the cGAS/STING pathway functions in chickens and that this is subverted by Fowlpox virus, a poxvirus that infects chickens, and that the mammalian CpG DNA sensing TLR9 pathway is functionally replaced in birds by TLR21:
3) Vaccinology
There are many fundamental questions about the development of immunological memory which remain poorly understood. The initial detection of a vaccine vector by the innate immune system has long lasting consequences for the development of memory responses. We use vaccinia virus, the vaccine that was developed to eradicate smallpox to help understand these fundamental mechanisms of vaccinology. For example we have developed a panel of mutant viruses which have differential impacts on the development of lymphocyte memory. We have defined the impact of removing individual and multiple immunomodulatory proteins from vaccinia virus on vaccination responses and the generation of CD8 T cell memory
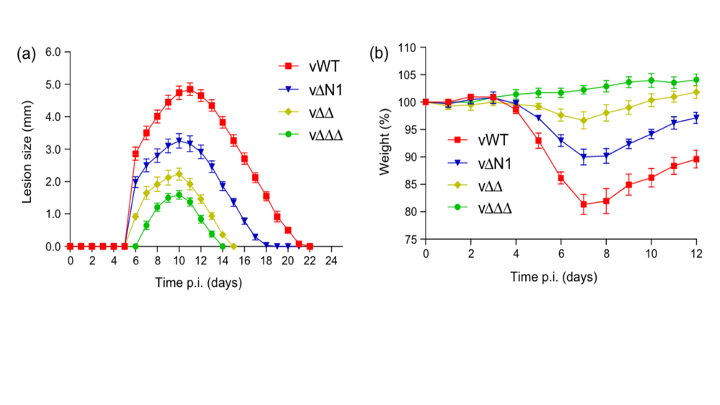
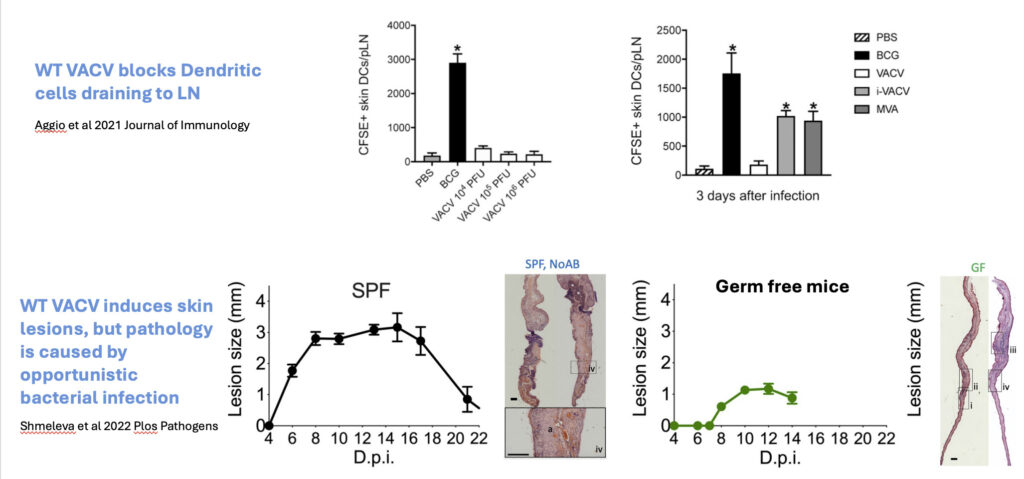
We are also working to understand the mechanisms by with vaccinia virus can generate such powerful memory immunity. We have recently discovered that vaccinia virus sabotages dendritic cell function and enters the draining lymph node to drive CD4 T cell activation, and that the pathology at the site of vaccination is almost completely dependent on opportunistic bacterial co-infection.
Collaborators:
Professor Daniel Mansur, University of Santa Catarina, Florianopolis, Brazil
Professor Geoffrey Smith, University of Oxford, UK
Professor Henning Walczak, University College London, UK
Professor Steve Jackson, Gurdon Institute, Cambridge, UK
Dr Antonio Rothfuchs, Karolinska Institute, Stockholm, Sweden
Dr Tim Halim, Cancer Research Institute, Cambridge, UK
Professor Alan Melcher, Institute of Cancer Research, London, UK
Professor Kevin Harrington, Institute of Cancer Research, London, UK
Dr Rodrigo Guabiraba, INRA, Tours, France
Professor Clare Bryant, University of Cambridge, UK
Dr Yorgo Modis, University of Cambridge, UK
Dr Amanda Chaplin, Leicester University
Dr James Edgar, University of Cambridge, UK
Dr Rahul Roychaudhuri, University of Cambridge, UK
Dr Nerea Irigoyen, University of Cambridge, UK
Dr Alexandre Belot, Inserm, Lyon, France
